Jean-Marc Taymans
Axes de recherche
LRRK2 & Parkinson
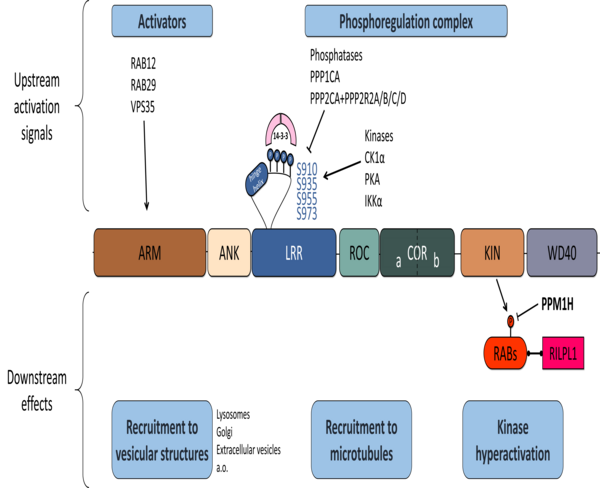
A major overall goal of our work is to develop strategies to disrupt LRRK2 complexes with its phosphoregulating partners as a therapeutic approach. Using structural, biophysical and biochemical approaches, we delimit binding interfaces between LRRK2 and its partners and protein-protein interaction modulators (PPIM) for the LRRK2:phosphatases complexes. Using cellular and in vivo methods, we then characterize how LRRK2 phosphorylation is affected by its phosphoregulation complex and validate PPIMs. For the most promising PPIMs, we proceed with validating their effect in cellular and in vivo models of disease. This approach is pursued in parallel with work (often done in collaboration) to develop and test agents that can modulate LRRK2 complexes as potential therapeutics.
A corollary of our hypothesis that the LRRK2 pathway is disturbed in disease is that LRRK2 related biomarker signatures would be altered in PD patients as well as in disease models. Therefore, a complementary aspect of our work is to study LRRK2 signatures, including LRRK2 pathway markers such as pS910/pS935-LRRK2, pS1292-LRRK2 or phosphorylation of Rab substrates, as well as omics signatures in patient cohorts and animal models. Recently, we found that S910 and S935 phosphorylation is reduced in urinary exosomes of PD patients (Taymans et al. NPJ PD 2023) and we also collaboratively determined proteomics signatures of LRRK2 activation in urine of LRRK2 PD patients as well as LRRK2 rat models (Vu et al. 2026).